09/06/2022 | pH | 8 MINUTE READ
What is Column Chromatography?

When working with solutions and chemicals in a laboratory setting, there are a wide range of techniques that can be used to obtain proper measurements. One such technique involves column chromatography, which is used to separate the various components that are present in a solution. If implemented in a chemistry lab, chromatography focuses on separating DNA, ions, and proteins.
Column chromatography can be administered for numerous applications, which include everything from food and beverage testing to forensic and drug testing. When performing column chromatography, it becomes possible to isolate components within a substance by adding an adsorbent to the column. The most common types of stationary phases that are used for column chromatography include alumina and silica gel. The mobile phases that are regularly used for column chromatography are organic solvents.
To understand what column chromatography is and how it’s useful for clean water, the primary aspect of chromatography involves the adsorption of the solutes that have been dissolved in the solution. Adsorption occurs with the assistance of the stationary phase mentioned previously. Once this process takes place, the solution will be separated into individual components.
The mobile phase and the component that needs to be properly isolated will be brought to the uppermost portion of the column, which results in the individual components moving at different rates. Components with less adsorption will move quicker, which allows them to be removed at a more rapid rate. This guide provides a comprehensive overview of column chromatography and its various applications.

Impact of pH on Chromatography
When performing column chromatography, one aspect of the process that can have a substantial impact on results is pH. The pH of the mobile phase can be altered depending on the needs you have, which makes it possible to control the selectivity, shape, and retention of the substance that’s being measured.
When performing reversed-phase liquid chromatography, the ionic strength and pH of the water-based portion of the mobile phase is essential to create more rugged measurement methods that aren’t sensitive to variations in conditions. As for ionic compounds, pH levels can change substantially. Controlling pH in a reversed-phase system will allow you to stabilize selectivity and retention. By keeping pH levels between 2-4, you’ll be able to maintain stable conditions.
The majority of silica-based columns are effective when pH levels vary from 2-8 pH. In the event that you’re using an HPLC column to separate carbohydrates and proteins, most of these polymer-based columns come with fantastic chemical stability and are effective regardless of where the pH levels are on the 1-14 range.

When performing a separation with column chromatography, make sure that you measure pH levels and properly adjust them within the aqueous component. This step should be done before the aqueous component is mixed with any organic modifiers. By changing pH levels beforehand, you should obtain precise results.
The pH level for the mobile phase that you use can directly affect the chromatography process in numerous ways. As mentioned previously, pH can affect peak shape, retention, and selectivity depending on what compound you’re currently analyzing. If you’re using a neutral or non-polar compound, pH changes won’t have much of an effect on retention and resolution.
On the other hand, ionizable compounds like bases and acids can be changed significantly in regards to selectivity and retention when pH levels are altered. A compound like benzoic acid can change by a considerable amount with retention when pH levels are shifted from three to seven. At a pH of 7.0, the benzoic acid will exist as an ionized benzoate anion, which is considered to be more ionic than benzoic acid at a 3.0 pH.
Column Chromatography Water Purification System Uses
Column chromatography requires purified water because of the sensitive applications it’s typically used with. Water is found in nearly every laboratory and is commonly used for the preparation of samples or as a reagent. Water can also be used when cleaning lab equipment and glassware in the facility.
When impurities are found in a solution that column chromatography is being used with, the results may be completely inaccurate. Liquid chromatography is considered to be highly sensitive, which is why it requires purified water. If there are high levels of contaminants in the water, the selectivity and sensitivity of the components can be substantially altered.

Keep in mind that different liquid chromatography applications require water for the preparation of eluents, for solid-phase extraction, and as reagent blanks. If you use impure and contaminated water for an extended period of time, drift and eventual degradation of the components may occur.
There are some specific impurities that are known to affect liquid chromatography as well as high-performance liquid chromatography (HPLC), the primary of which are organic compounds. Contaminants like particulates, ions, and bacteria can also adversely affect these chromatography techniques.
Any organic impurities that are found in the water may result in several chromatography issues. For one, the analyte that’s currently being measured can compete directly with the impurities located in the stationary phase. Large peaks may also be formed. It’s possible that the impurities will produce ions that consist of masses close to the primary elements that affect the efficiency of ionization.
In the event that water contains inorganic compounds and is paired with ion chromatography, the reproducibility and sensitivity might be affected. The existence of these impurities could result in drift, enlarged peaks, and ample background noise, which lead to less accurate results. As for particulates and bacteria, these contaminants may block components and produce inaccurate results.
Food Testing
In the past, food testing was only possible when performing these tests with raw samples. With the introduction of high-performance liquid chromatography, lab technicians were able to combine this technology with mass spectroscopy to start testing processed foods. Through these tests, it was discovered that the types of meat in food preparation were commonly sheep or beef.

Beverage Testing
It’s also possible to use chromatography to test beverages. The beverage industry uses the standard chromatography technique to ensure that their products have a consistent taste that consumers can rely on.
Drug Testing
Chromatography is highly effective when used in drug testing. This technique is able to separate any type of drug from the bloodstream, which makes it easy to determine if an individual has recently consumed drugs. Chromatography is regularly used for drug testing in sports like football and baseball.
Forensic Testing
The chromatography technique has long been used for forensic testing to ensure that criminals are caught even when evidence is minimal. Cloth and blood samples are analyzed with chromatography to identify criminals.

How Do We Test pH?
There are many effective meters and sensors that can be used to test pH levels in a solution. When preparing solutions, precise measurements are needed to ensure that the composition of the solution is exact. You can select a benchtop pH meter package to receive all of the tools you need to perform pH measurements in the lab. This type of kit consists of a pH sensor, benchtop meter, and buffer solutions.
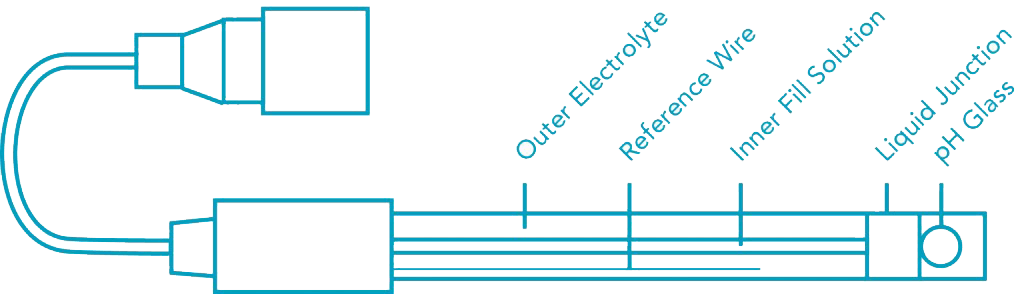
How Does a pH Sensor Work?
The right pH sensor can provide you with continuous measurement due to the inclusion of a low-flow pH cell that’s comprised of a laboratory grade electrode. This flow cell is ideal when used with high-performance liquid chromatography.
It’s also possible to use these cells in all applications that require continuous pH monitoring in relatively low flows. Keep in mind that there are different capacities you can choose from. You could either select a 50-microliter flow cell or a 200-microliter flow cell.
The Three Types of Advanced Water Quality Analytics Instruments
When you need to analyze the quality of water, there are three distinct types of advanced water quality analytics instruments that you can choose from, which include:
- Inductively coupled plasma mass spectrometry
- Gas chromatography mass spectrometry
- Liquid chromatography mass spectrometry
By using one or more of these instruments, it’s possible for you to identify and analyze various contaminants, pollutants, and trace metals. For instance, solvents and pharmaceuticals could be identified with one of the aforementioned instruments. The reason why these instruments are highly effective is because they are able to detect these contaminants even when they are present in low concentrations.
Once you’ve performed water quality tests and measurements with these tools, you might want to use an information management system to make sure that every piece of data captured in the lab is properly stored and maintained.
Column chromatography is highly effective at separating chemical compounds with the help of a stationary phase and mobile phase. Regardless of the intended application, chromatography has proven to be useful for food and beverage testing as well as drug testing. However, chromatography requires purified water to ensure that the results from these tests are accurate, which is why it’s recommended that you regularly test pH levels before conducting chromatography experiments.
Posted by Dominic O'Donnell on September 6, 2022
Sensorex is a global leader in the design and manufacture of quality sensors for water quality and process applications. The company offers more than 2000 sensor packages for pH, ORP, conductivity, dissolved oxygen, free chlorine, chlorine dioxide, UV transmittance and other specialty measurements, as well as a full line of sensor accessories and transmitters. Its expert technical support engineers solve analytical sensor challenges with custom designs and off the shelf products.




